
Worth knowing about Novel food
If you are going to sell new food products that have never been produced before, or that have been produced in completely new ways, you need to check whether or not your product or process requires Novel Food approval from the EU.
What is Novel Food?
"Novel food" is a term used by the EU in respect of approving "radically new food". Novel Food or "new food" approvals are EU measures that are designed to protect consumers from the potentially dangerous effects of unknown foods.
What do I need Novel Food approval for?
If your product is brand new, or has been produced using a completely new process, it may require Novel Food approval from the EU before it can be sold on the European market.
The six processing technologies on this website, high pressure, microwaves, pulsed electric fields, ultrasound, UV-C light and plasma-activated water, have been approved in varying degrees as being acceptable processes in the production of different food products. Some of these were introduced to the market before 1997 and thus do not require this approval (for example, microwave products). Plasma-activated water is still in the laboratory stage and has not yet been approved for use with food. Some products have received Novel Food approval, such as UV-treated mushrooms, yeast, bread, milk and fruit that has undergone high-pressure treatment. The full list can be viewed here:
EUR-Lex - 32017R2470 - EN - EUR-Lex (europa.eu)
Novel Food approval has often been used in conjunction with patents in order to build the value of new products. Furthermore, Novel Food products often come close to being categorised as health foods and thus it has been important for those involved to see the EFSA-approved health claims.
Norwegian Novel food products
The Scandinavian countries have been particularly forward-looking when it comes to Novel Food and there are as many as seven Norwegian products among the first ones to have been approved. Examples of this include krill, 2x vitamin K2, beta glucan from mushrooms, squid oil, Calanis finmarchicus and peptides from shrimp shells. Otherwise, the most well-known Novel Food products are chia seeds and noni juice.
As regards new technologies or new combination processes, these are less commonly found among Novel Food applications. However, we do have some examples of this, e.g. UV irradiation of fungi and other products that results in products enriched with vitamin D, and high-pressure processing of juices and jams.
Where can I obtain help for assessing an application?
The Norwegian Food Safety Authority in Norway has very good general information with links to updated Novel Food approvals in the EU. The Norwegian Food Safety Authority also has several people who have been involved in coordinating the work undertaken by the European Commission in this field.
Nofima also has professionals with experience of Novel Food processes and there are also some consultancy companies in Europe that specialise in helping the industry in this respect. It should be entirely possible to obtain such approvals, even for the food industry, and much of the job often involves obtaining documentation to show that the product in question does not fall under the Novel Food Regulations.
History of Novel Food approval in Europe
In Europe, Novel Food is a term used for food and drink introduced on the market after 15 May 1997. Inspired by approval schemes for pharmaceuticals, the EU established advanced methods for toxicological and microbiological safety for new food products. This resulted in additional requirements for completely new imported foods and for foods produced using completely new processes.
EU Novel Food approval is based on transparency and accountability, but has often proved to be a demanding process in terms of time, resources and costs for the food industry. Before the scheme was simplified, it took an average of almost four years to obtain approval for a full application. On the other hand, approval of applications for permission to sell copies of previously approved Novel Food products only took a few months, and thus most of the applications received fell into this category.
During the first 20 years, only 128 new products were approved, while 523 copy applications were submitted. The fact that free-passenger strategies have often been more profitable than being the first on the scene has been detrimental for innovations in the food industry. The process involved companies initially having to obtain approval from their own national food authorities before the European Food Safety Authority (EFSA) could comment on food safety.
In 2018, the European Commission reviewed the scheme and this resulted in a new scheme that is supposed to be simpler, faster and more transparent. Companies can now apply directly to the EU without first having to obtain the approval of their national food authorities.
The new scheme provided applicants with better protection. Applicant companies can now tick off data protection and exclusivity, but exclusivity only applies if the process is still protected by secrecy. Data protection with individual authorisation for applicant companies should apply for five years, otherwise generic authorisation will be granted. Nevertheless, we have noticed that companies are still wanting to patent processes for which approval has been applied for.
After 2018, the European Commission also created a so-called "Union List"of all previously approved Novel Food products so that the food industry does not need copy applications in order to produce or distribute these new products. If no data protection has been applied for and approved under the new regulations, all approved Novel Food products will appear on the "Union List". Half of the products for which EU approval is currently being applied for are such products.
After the new scheme came into force, almost a hundred applications were received during the first year, compared to the norm of ten applications annually.
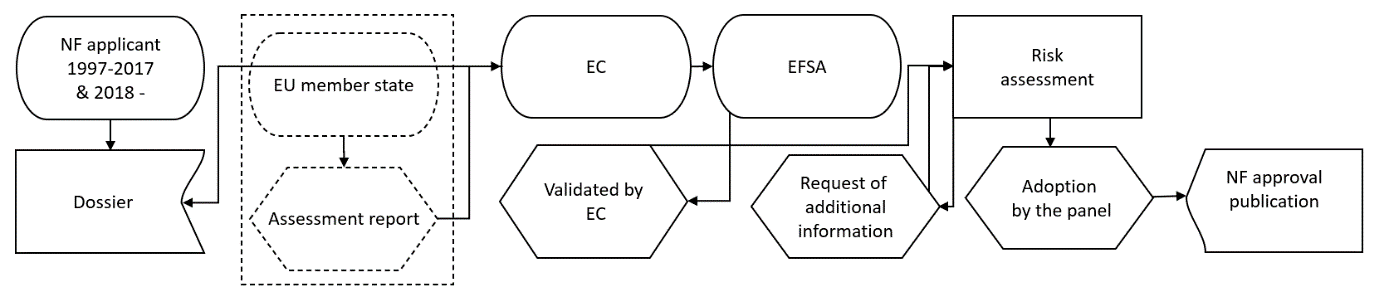
The figure above illustrates new and old approvals for new food in Europe. During the first 20 years, each EU member state was involved in the process, something which delayed the approval period.
Research on Novel Food
Under our iNOBox research project we have studied the EU's Novel Food Regulations, especially those relating to cooperation and patenting. Two scientific papers have been published under innovation in the British Food Journal. These can be found in the reference list below.
Links and resources
- The Norwegian Food Safety Authority’s website on Novel Food
- EU Novel Food information
- European Food Safety Authority (EFSA) information on Novel Food
- The EU's Novel Food Catalogue contains a guide to all the products derived from the animal and plant kingdom that are covered by the Novel Food Regulations.
- A series of questions and answers about legislation relating to Novel Food: Q&As (europa.eu)
| Title | Author(s) | Year | |
|---|---|---|---|
| Radical innovation in a conservative industry. Selected cases of new product development in the Norwegian food industry. | Sveinung Grimsby | 2021 | Go to publication |
| New novel food regulation and collaboration for innovation | Sveinung Grimsby | 2020 | Go to publication |
| European novel food, patents and brokers of knowledge | Sveinung Grimsby, Magnus Gulbrandsen | 2022 | Go to publication |
